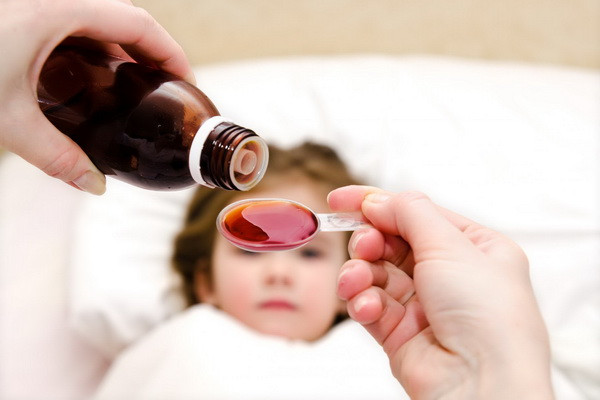
The toxic substance ethylene glycol was found in a batch of Doc-1 Max syrup
Tashkent, Uzbekistan (UzDaily.com) -- In Samarkand, several children were hospitalized as a result of side effects of the drug Dok-1 Max. On the basis of the relevant order of the Ministry of Health, members of the working group conducted an official investigation on the fact with a site visit. This was reported by the press service of the ministry.
Tablets and syrup Doc-1 Max manufactured by the Indian company Marion Biotech Pvt. Ltd in our country were registered in 2012 and went on sale in the same year.
According to the Agency for the Development of the Pharmaceutical Industry, each drug series was tested with the subsequent issuance of a certificate of conformity. This drug was imported into our country by Quramax Medical LLC.
To date, 18 out of 21 children with acute respiratory disease have died as a result of taking Doc-1 Max syrup.
It was found that the deceased children, before admission to hospital treatment, took this drug at home for 2-7 days 3-4 times a day, 2.5-5 ml, which exceeds the standard dose of the drug for children.
All children were given the drug without a doctor’s prescription. Since the main component of the drug is paracetamol, Doc-1 Max syrup was incorrectly used by parents as an anti-cold remedy on their own or on the recommendation of pharmacy sellers. And this was the reason for the deterioration of the condition of patients.
In fact, paracetamol should only be used at a body temperature of 38-38.5 C and above 1 or 2 times a day, 100-125 mg for a child under the age of one year, 200 mg for children aged 1-3 years and 250 mg - from 3 to 5 years. At normal body temperature, taking this drug is strictly prohibited.
Preliminary laboratory studies have shown that this series of Doc-1 Max syrup contains ethylene glycol. This substance is toxic, and about 1-2 ml/kg of a 95% concentrated solution can cause serious changes in the patient’s health, such as vomiting, fainting, convulsions, cardiovascular problems and acute kidney failure.
Taking into account all cases of violation of the law identified by the working group of the Ministry during the study, 7 responsible employees were dismissed from their positions due to the fact that they were negligent and inattentive to their duties, did not analyze child mortality in a timely manner and did not take the necessary measures, also disciplinary measures were applied to a number of specialists.
The collected documents were handed over to law enforcement agencies.
Currently, tablets and syrups of the drug Doc-1 Max are withdrawn from sale in all pharmacies of the country in the prescribed manner. The shortcomings identified on the basis of the study materials, the issue of the responsibility of medical workers will be considered at a separate meeting of the Collegium of the Ministry of Health.